Protocol for a randomised controlled feasibility trial of exercise
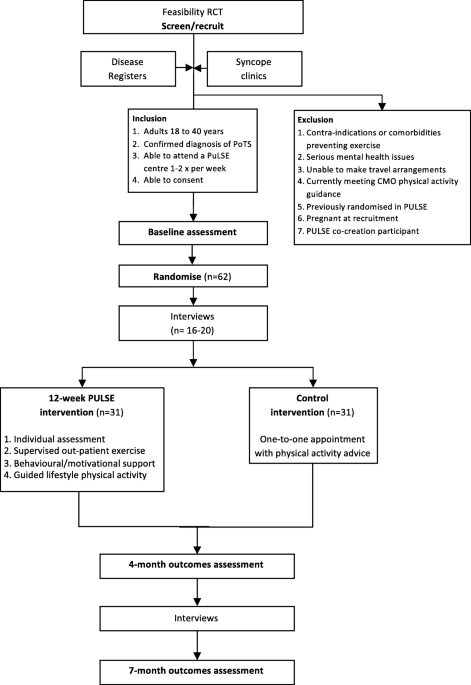
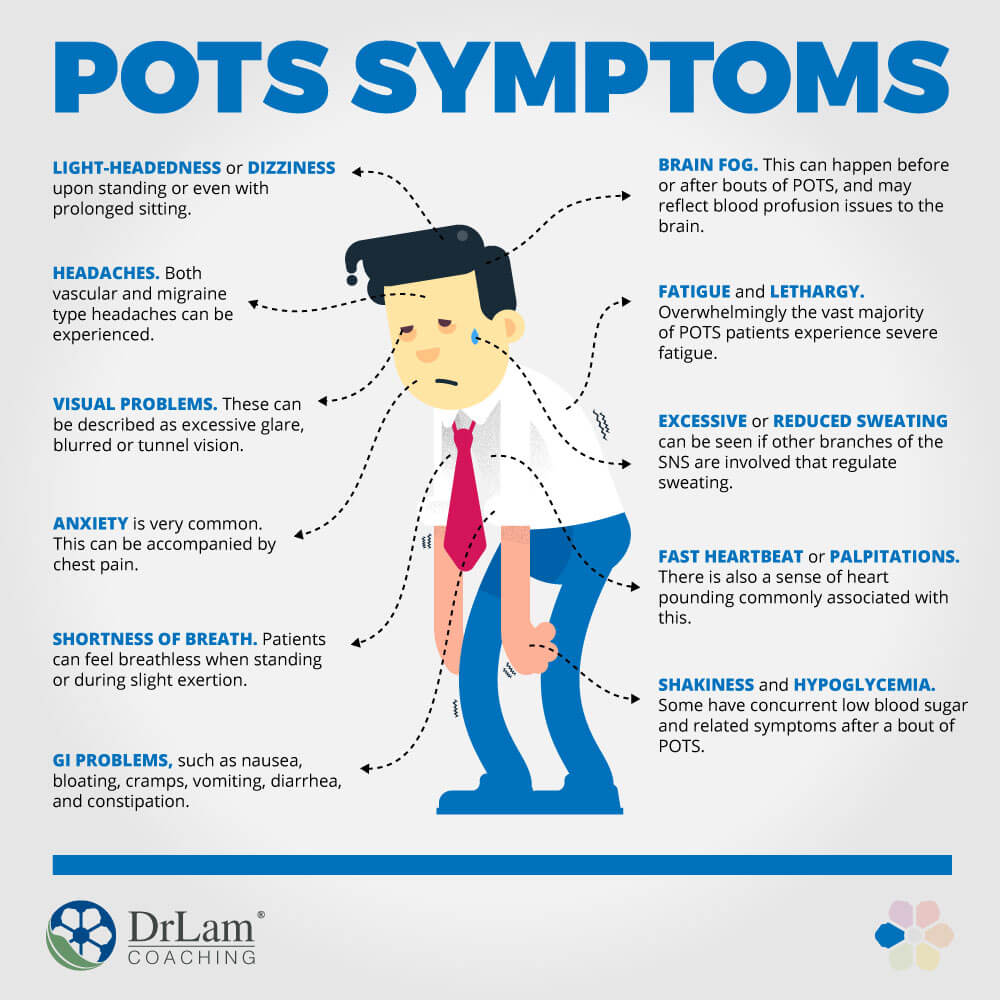
Background Postural orthostatic tachycardia syndrome (POTS) is an autonomic nervous system disorder causing an abnormal cardiovascular response to upright posture. It affects around 0.2% of the population, most commonly women aged 13 to 50 years. POTS can be debilitating; prolonged episodes of pre-syncope and fatigue can severely affect activities of daily living and health-related quality of life (HRQoL). Medical treatment is limited and not supported by randomised controlled trial (RCT) evidence. Lifestyle interventions are first-line treatment, including increased fluid and salt intake, compression tights and isometric counter-pressure manoeuvres to prevent fainting. Observational studies and small RCTs suggest exercise training may improve symptoms and HRQoL in POTS, but evidence quality is low. Methods Sixty-two people (aged 18–40 years) with a confirmed diagnosis of POTS will be invited to enrol on a feasibility RCT with embedded qualitative study. The primary outcome will be feasibility; process-related measures will include the number of people eligible, recruited, randomised and withdrawn, along with indicators of exercise programme adherence and acceptability. Secondary physiological, clinical and health-related outcomes including sub-maximal recumbent bike exercise test, active stand test and HRQoL will be measured at 4 and 7 months post-randomisation by researchers blinded to treatment allocation. The PostUraL tachycardia Syndrome Exercise (PULSE) intervention consists of (1) individual assessment; (2) 12-week, once to twice-weekly, supervised out-patient exercise training; (3) behavioural and motivational support; and (4) guided lifestyle physical activity. The control intervention will be best-practice usual care with a single 30-min, one-to-one practitioner appointment, and general advice on safe and effective physical activity. For the embedded qualitative study, participants (n = 10 intervention, n = 10 control) will be interviewed at baseline and 4 months post-randomisation to assess acceptability and the feasibility of progressing to a definitive trial. Discussion There is very little high-quality research investigating exercise rehabilitation for people with POTS. The PULSE study will be the first randomised trial to assess the feasibility of conducting a definitive multicentre RCT testing supervised exercise rehabilitation with behavioural and motivational support, compared to best-practice usual care, for people with POTS. Trial registration ISRCTN45323485 registered on 7 April 2020.

PDF) Protocol update for a randomised controlled feasibility trial of exercise rehabilitation for people with postural tachycardia syndrome: the PULSE study

A study protocol for a randomised controlled feasibility trial of an intervention to increase activity and reduce sedentary behaviour in people with severe mental illness: walking fOR Health (WORtH) Study.
What are Pilot Studies and Clinical Trials?

Lifestyle and Behavioral Interventions to Reduce the Risk of Metabolic Syndrome in Community-Dwelling Adults With Serious Mental Illness: Implications for Nursing Practice

Exercise as medicine – evidence for prescribing exercise as therapy in 26 different chronic diseases - Pedersen - 2015 - Scandinavian Journal of Medicine & Science in Sports - Wiley Online Library

Protocol for a randomised controlled feasibility trial of exercise rehabilitation for people with postural tachycardia syndrome: the PULSE study, Pilot and Feasibility Studies
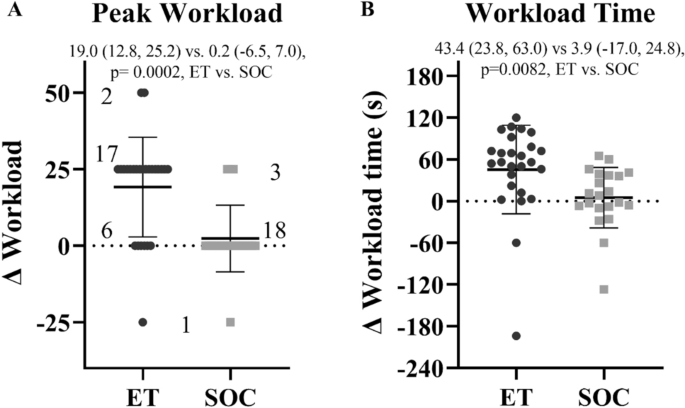
Semi-supervised exercise training program more effective for individuals with postural orthostatic tachycardia syndrome in randomized controlled trial

Postural Tachycardia Syndrome (POTS)

Arryhthmias - ACPICR

Loop Nikki Holliday

Nikki Holliday - Design Manager - Coventry University

Sandeep PANIKKER, Honorary Consultant Cardiologist and Electrophysiologist, Doctor of Philosophy, Imperial College London, London, Imperial, National Heart and Lung Institute

Volume 6, issue 1 Pilot and Feasibility Studies

PDF] Endurance Exercise Training in Orthostatic Intolerance: A Randomized, Controlled Trial

Historical Background