What is the change in internal energy (in J) of a system that absorbs 0.464 kJ of heat from its surroundings and has 0.630 kcal of work done on it?

I found an increase of 3100J Have a look

Solved Be sure to answer all parts. What is the change in

A system absorbs `600J` of heat and work equivalent to `300J` on

Solved Be sure to answer all parts. What is the change in

The elastic properties, elastic models and elastic perspectives of

Solved: Chapter 6 Problem 11P Solution
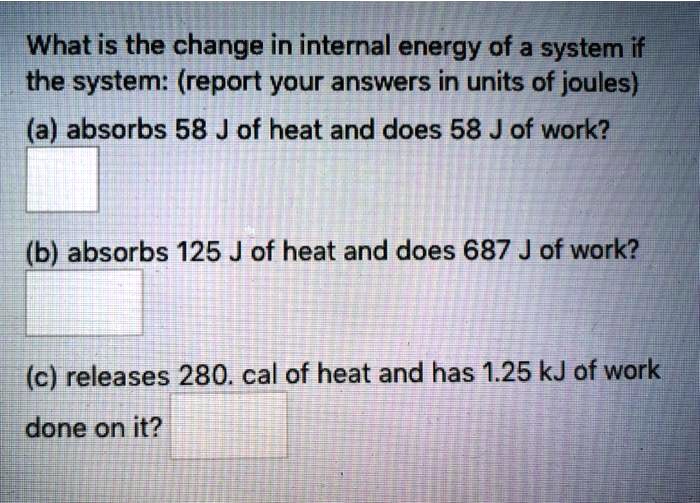
SOLVED: What is the change in internal energy of a system if the

ME532 AdvancedHT IIConvectionandMassTransfer PDF, PDF

Handbook On Energy Conscious Buildings, PDF, Humidity

SOLVED: 1.3) A system releases 125 kJ of heat while 104 KJ of work
You may also like
Related products









