Real gases


Gases Real Gases

Deviation of real gas from ideal behaviour

1.5 Real Gases and the Virial Equation - Mail
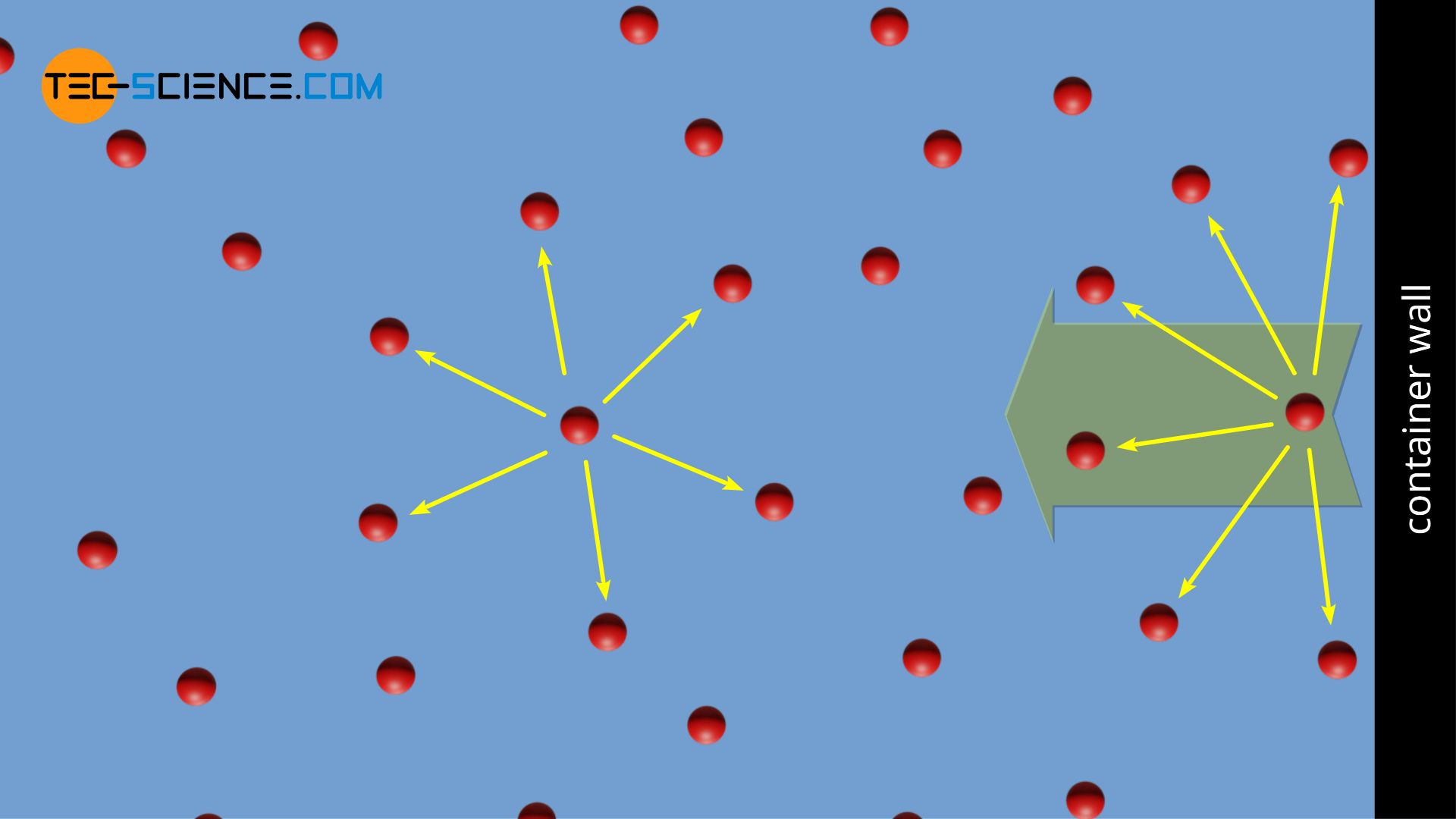
Van der Waals equation (gas law for real gases) - tec-science

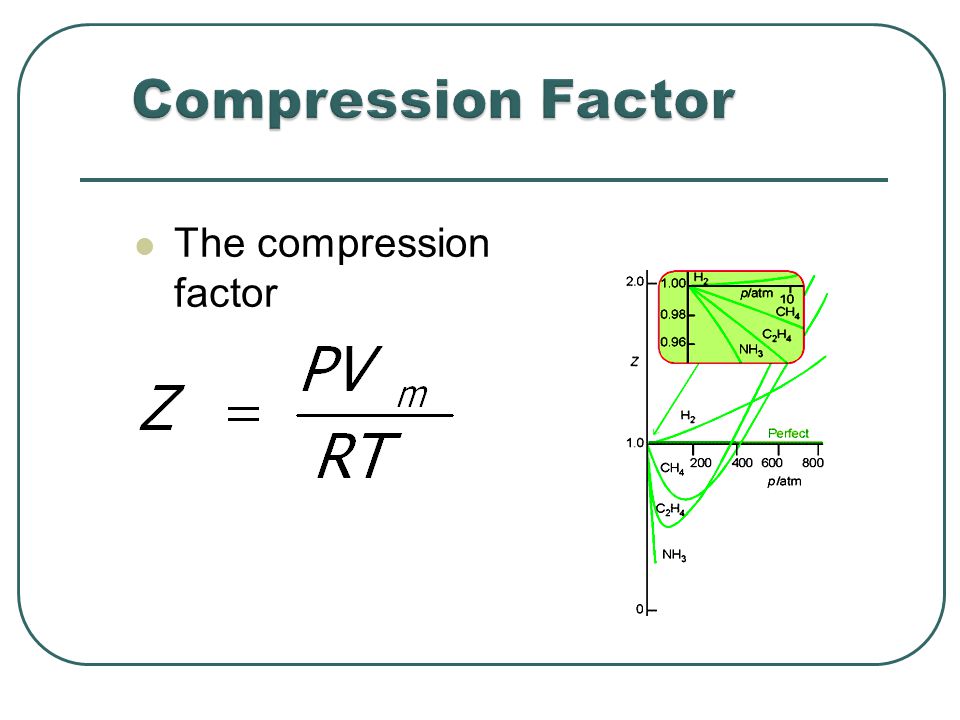
Description of real gases: Compression factor
Solved] Real gases behave differently than ideal gases when conditions make

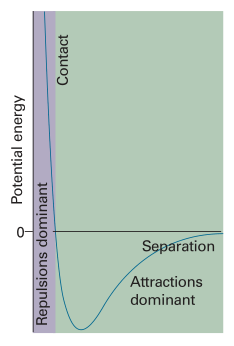
Real Gas vs Ideal Gas

Real Gases vs Ideal Gases & the Compressibility Factor

17 Behaviour Gases Royalty-Free Images, Stock Photos & Pictures
Chemistry: Real gases

Ideal Gases & Real Gases Deviations from Ideal Behavior : r/Mcat
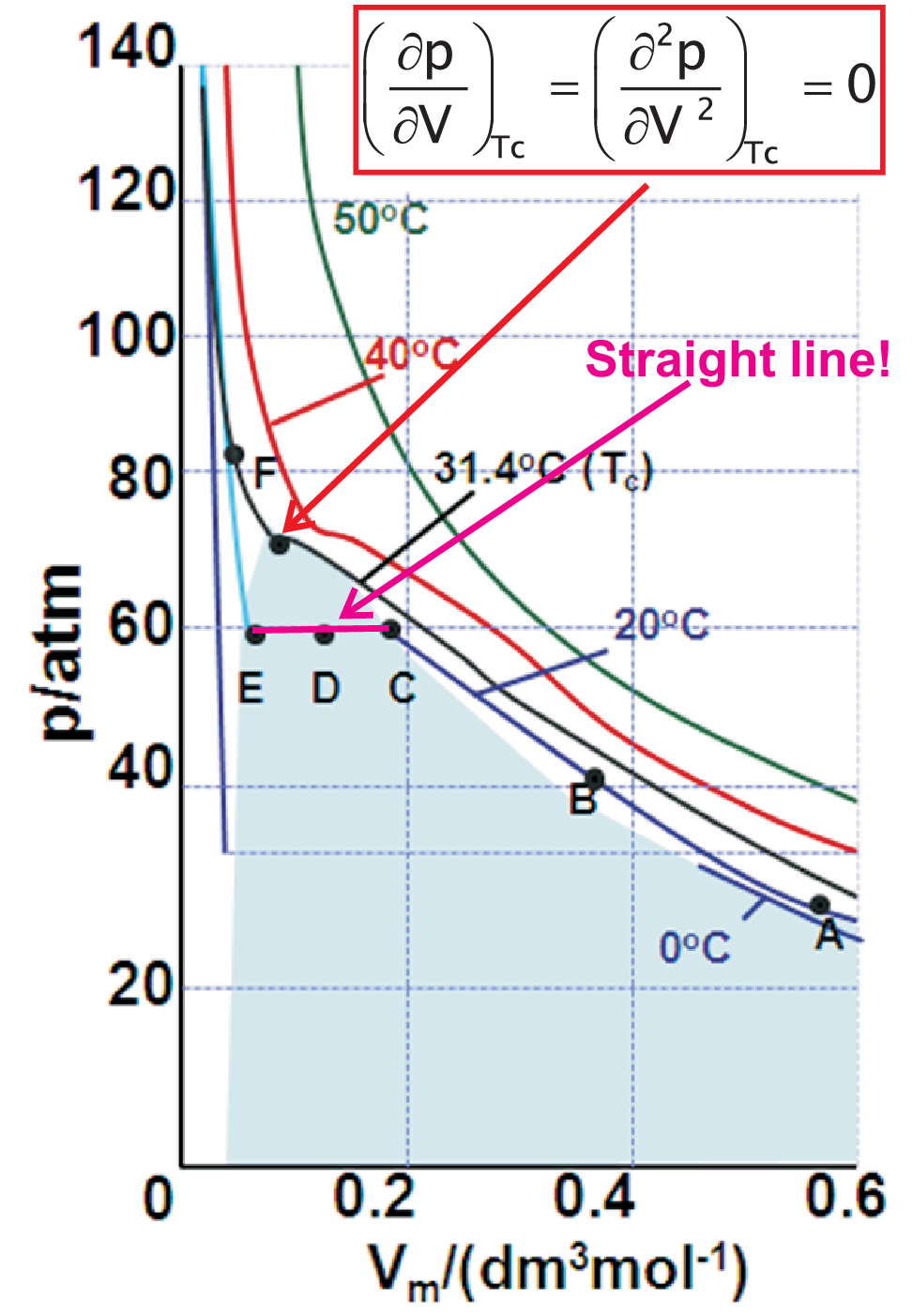
Condensation of real gases

The given graph represents the variation of Z (compressibility factor = \[\dfrac{{PV}}{{nRT}}\] ) versus P, for three real gases A, B and C. Identify the only incorrect statement.
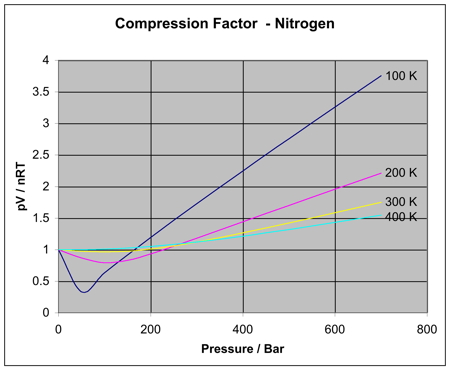
For real gases, how does a change in pressure affect the ratio of PV to nRT?